Tag: generic drugs

Excipients in generics: how inactive ingredients influence tolerance
Excipients in generic drugs-though labeled 'inactive'-can trigger allergic reactions, digestive issues, and intolerances. Learn how fillers, dyes, and preservatives in generics affect tolerance and what to do if you suspect a reaction.

Effective Patent Life: Why Market Exclusivity for Drugs Is Shorter Than You Think
Effective patent life for drugs is often only 10-15 years, not 20, because patents start ticking before approval. Regulatory delays, secondary patents, and exclusivities shape how long a drug stays exclusive.
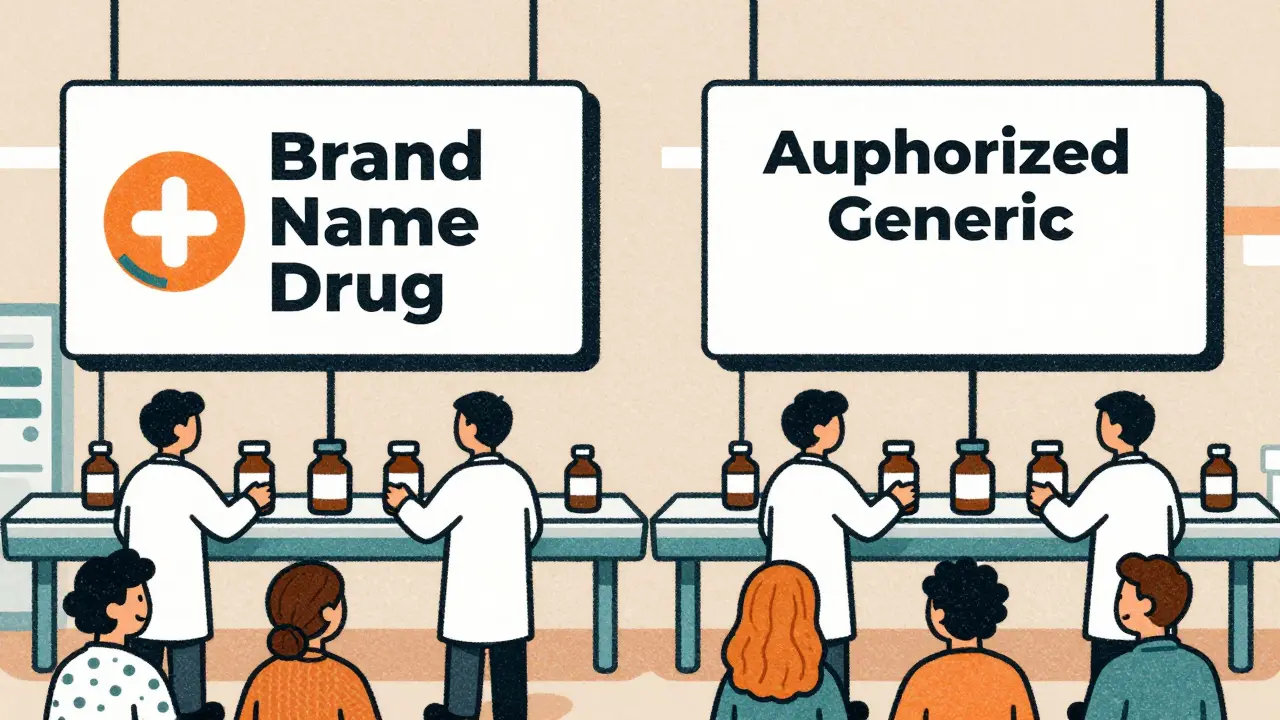
Future Role of Authorized Generics: Market Outlook
Authorized generics let brand drugmakers sell their own drugs under generic labels after patent expiry. With new FDA rules and rising biosimilar competition, their role is shifting from market control to managed transition-impacting prices, access, and competition.

Nitrosamine Contamination in Generic Drugs: Recent Recalls and Regulatory Shifts
Nitrosamine contamination in generic drugs has triggered over 500 FDA recalls since 2018. Learn which medications were affected, how regulators responded, and what it means for your health and access to affordable medicine.

Workers' Compensation and Generic Substitution: What You Need to Know in 2026
Generic substitution in workers' compensation saves billions without sacrificing care. Learn how FDA-approved generics replaced brand-name drugs in 2026, state laws, provider challenges, and why this isn't just about cost-it's about smart medicine.
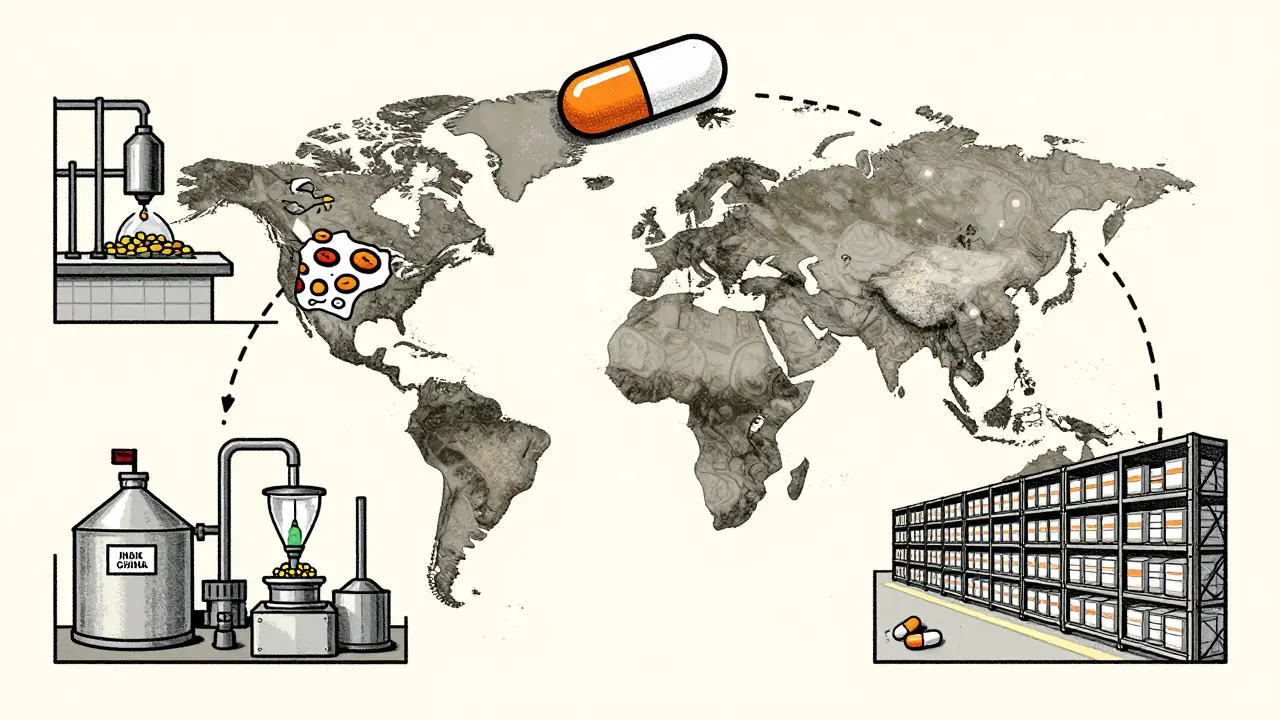
Quality concerns: when clinicians question generic manufacturing
Clinicians are raising alarms about the quality of generic drugs made overseas, citing higher rates of adverse events and supply chain risks. Here’s what’s really happening behind the label.